What is elimination reaction with example?
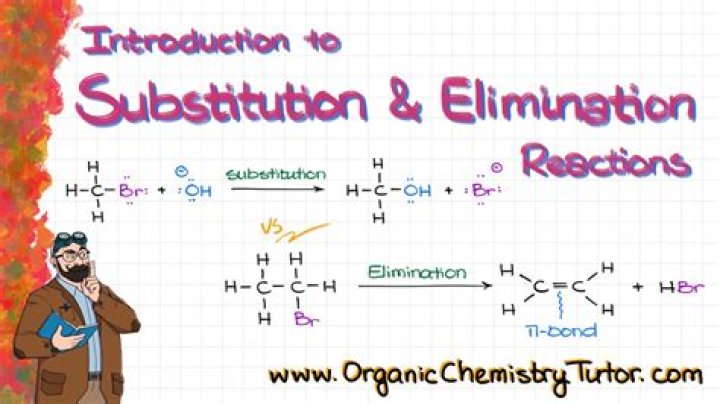
Elimination reactions are commonly known by the kind of atoms or groups of atoms leaving the molecule. The removal of a hydrogen atom and a halogen atom, for example, is known as dehydrohalogenation; when both leaving atoms are halogens, the reaction is known as dehalogenation.
What are the two preparations by elimination?
The most common elimination reactions are dehydrohalogenation and dehydration.
How are alkenes formed from alkanes?
An alkene represents an unsaturated hydrocarbon with double bonds, while an alkane is a saturated hydrocarbon with only single bonds. To convert an alkane to an alkene, requires that you remove hydrogen from the alkane molecule at extremely high temperatures. This process is known as dehydrogenation.
Do alkenes undergo elimination reactions?
Elimination reactions form alkenes as well as alkynes. Alkyne-forming elimination reactions are described in a subsequent section. Common Features of Elimination Reactions (9.1A) A variety of different types of substrates undergo elimination reactions to form alkenes, but many of these reactions have common features.
Why does elimination reaction occur?
The reaction usually occurs in the complete absence of a base or the presence of only a weak base (acidic conditions and high temperature).
What are elimination reactions give two examples?
The E2 and E1 mechanisms differ in the timing of bond cleavage and bond formation, which is analogous to the SN1 and SN2 reactions….E1 vs. E2 Reaction.
| E1 Reaction | E2 Reaction | |
|---|---|---|
| Examples of substrate | 2-methyl-2-butanol and 2-methyl-2-butene | 2-chloro-2-methyl butane and 1-propanol |
Do Alkenes undergo elimination reactions?
What is the main reaction of Alkenes?
The most common type of reaction for alkene is the addition reaction to C=C double bond. In addition reaction, a small molecule is added to multiple bond and one π bond is converted to two σ bonds (unsaturation degree decreases) as a result of addition. Addition reaction is the opposite process to elimination.
How are alkenes formed in elimination reactions?
In many elimination reactions that give alkenes, A (or B) is an H atom. In this reaction, the loss of both an H and the leaving group L from adjacent C atoms leads to the formation of the C=C bond. Typically H is removed as a proton (H+) by a base, and L departs with its bonding electron pair as -:L.
How do you determine the stability of an alkene?
Stability of Alkenes Alkenes are reduced to alkanes by the action of hydrogen gas in the presence of a catalyst. The exothermicity of this reaction is called the heat of hydrogenation, and these values give information about the stability of the alkene.
What is the molecular weight of a double bond in alkenes?
Structure and Synthesis of Alkenes Alkenes (olefins) are hydrocarbons which have carbon–carbon double bonds. A double bond is a bond and a bond. Double bond B.D.E. = 146 kcal/mol bond B.D.E. = 83 kcal/mol Therefore B.D.E. must = 63 kcal/mol. A bond is weaker than a bond.
What is the nomenclature of alkenes?
Nomenclature of Alkenes Simple alkenes are named like alkanes (root from the longest carbon chain), but the –anesuffix is replaced by-ene. When the chain is longer than 3 carbons, number the atoms such that the double bondis given the lowest number (i.e. start at the end nearest the double bond).