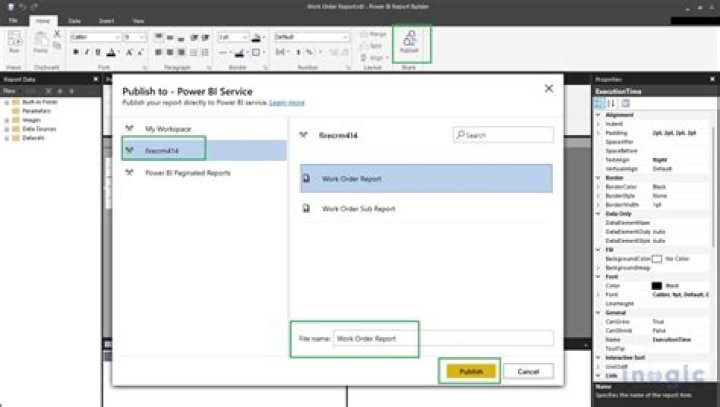
What type of reaction is rusting of iron?
oxidation reaction
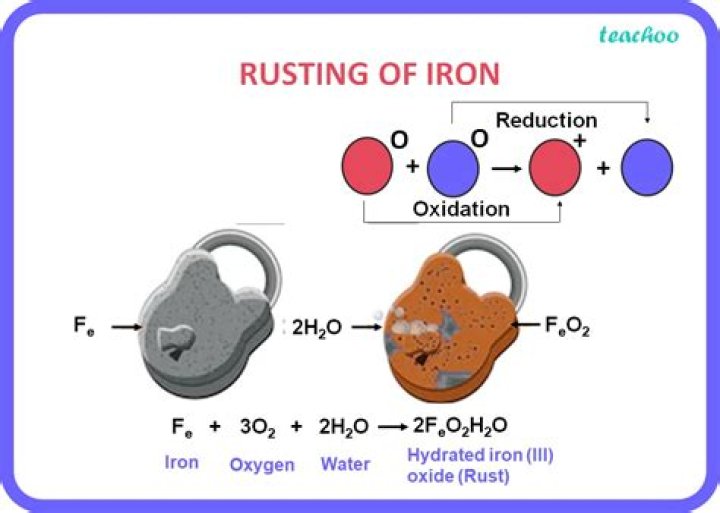
Rusting is an oxidation reaction. The iron reacts with water and oxygen to form hydrated iron(III) oxide, which we see as rust.
What is iron rusting an example of?
Rust is clearly a substance that is different from iron. Rusting is an example of a chemical change. A chemical property describes the ability of a substance to undergo a specific chemical change. A chemical property of iron is that it is capable of combining with oxygen to form iron oxide, the chemical name of rust.
Is rusting of iron a decomposition reaction?
Option A, Combination reaction is the correct answer. Rust is an iron oxide. Rust is the product of a combination reaction of iron with oxygen: Since this new compound is produced or formed from a metal and a non-metal, it is termed as an ionic compound.
What type of reaction is rusting and combustion?
Corrosion and combustion are both examples of oxidation reactions involving substances reacting with oxygen.
Is rusting of iron combustion?
No. Rusting is not a combustion reaction. The process of rusting is called oxidation reaction, in which there is a gain of oxygen with the loss of electrons. Combustion is an exothermic redox chemical reaction.
Is rusting of iron an exothermic reaction?
Iron rusting is an exothermic reaction. Iron rusting is an exothermic reaction. How much energy is released when 2.79 g of iron reacts with excess oxygen? As always the first step is to convert the given quantity (2.79 g Fe) into moles.
Is rusting of iron a combustion reaction?
Is rusting double decomposition reaction?
Explanation: Because it is an example of redox reaction.
Is rusting of iron a type of combustion?
Is rusting of iron An example of combustion?
Yes, rusting of iron is an example of slow combustion.
Is rust a type of combustion?
Rust forms when iron and oxygen react in the presence of water or moisture in the air. The process of rusting is a combustion reaction, similar to fire. Left in contact with oxygen, iron will react with the oxygen to form rust.
Is corrosion a combustion reaction?
The combination of a metal with oxygen or other chemical to produce a metal compound is called corrosion. Combustion is like corrosion in that it involves combining a substance with oxygen, but this time very quickly to produce heat and light as a flame.
What type of chemical reaction is rusting of iron?
Chemistry of Rusting of Iron Corrosion of any metal is a natural process. It results in the conversion of the metal into its chemically stable form that can be either oxides or hydroxides or sulphides. Rusting is a type of Chemical Change. It results in the formation of Iron Oxide which is an entirely new substance.
What is the formation of rust?
The formation of rust, that is a mixture of iron oxides, on the surface of iron objects or structures. The rust is formed by the redox reaction between oxygen and iron in an environment containing water such as air containing high levels of moisture.
Is rust an oxidation or an oxidization reaction?
Although rust is considered the result of an oxidation reaction, it’s worth noting not all iron oxides are rust. Rust forms when oxygen reacts with iron but simply putting iron and oxygen together isn’t sufficient. Although about 20 percent of air consists of oxygen, rusting doesn’t occur in dry air. It occurs in moist air and in water.
What is the reaction between iron oxide and hydroxide?
The iron ion and the hydroxide ion react to form iron hydroxide: 2Fe 2+ + 4OH – → 2Fe (OH) 2 The iron oxide reacts with oxygen to yield red rust, Fe 2 O 3.H 2 O Because of the electrochemical nature of the reaction, dissolved electrolytes in water aid the reaction.