What is the electrophile in nitration?
The electrophile is the “nitronium ion” or the “nitryl cation”, NO+2. This is formed by reaction between the nitric acid and the sulphuric acid.
Which electrophile is responsible for nitration?
The active electrophile for nitration is widely believed to be the nitronium ion (NO2 +), and the putative mechanism for the generic reaction is outlined in Fig. 1 [8].
How do you form a no2 electrophile?
The formation of the electrophile The electrophile is the “nitronium ion” or the “nitryl cation”, NO2+. This is formed by reaction between the nitric acid and the sulphuric acid.
How is the electrophile generated in the nitration of benzene?
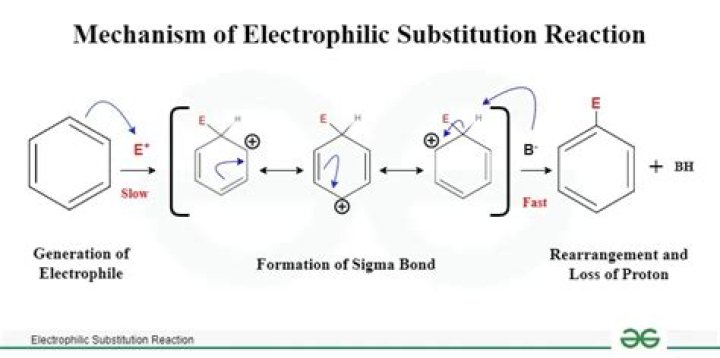
The arenium ion loses a proton and forms nitrobenzene. Hence, the generation of electrophile in nitration of benzene takes place by protonation of nitric acid with the help of sulphuric acid. The arenium ion is the resonance stabilized carbocation which forms during the electrophilic substitution in an aromatic ring.
Which is most readily nitrated?
Phenol is more easily nitrated than benzene.
What is the responsible for nitration reaction?
H2SO4 is responsible for the generation of nitronium ion (NO+2) which is responsible for nitration.
How aniline is nitrated?
In case of nitration in aniline, the Nitric Acid protonates the aniline to form the Anilinium ion. Now since the nitrogen atom has no lone pair to conjugate, it has no mesomeric effect on the ring, the but as nitrogen is now protonated, it has a high negative inductive effect.
What is the electrophile in the acylation of benzene?
The electrophile is CH3CO+. It is formed by reaction between the ethanoyl chloride and the aluminium chloride catalyst. The hydrogen is removed by the AlCl4- ion which was formed at the same time as the CH3CO+ electrophile.
How do you do acylation?
The Friedel-Crafts acylation reaction involves the addition of an acyl group to an aromatic ring. Typically, this is done by employing an acid chloride (R-(C=O)-Cl) and a Lewis acid catalyst such as AlCl3. In a Friedel-Crafts acylation reaction, the aromatic ring is transformed into a ketone.
Which reagent is used in acylation?
The two reagents used for acylation of benzene are: CH3COCl (acetyl chloride) and anhydrous AlCl. (CH3CO)2O (acetic anhydride) and anhydrous AlCl.
Which is most readily nitrated phenol or aniline?
Answer: Aniline is more easily nitrated than phenol as the NH2 group is a stronger R showing group than OH.

