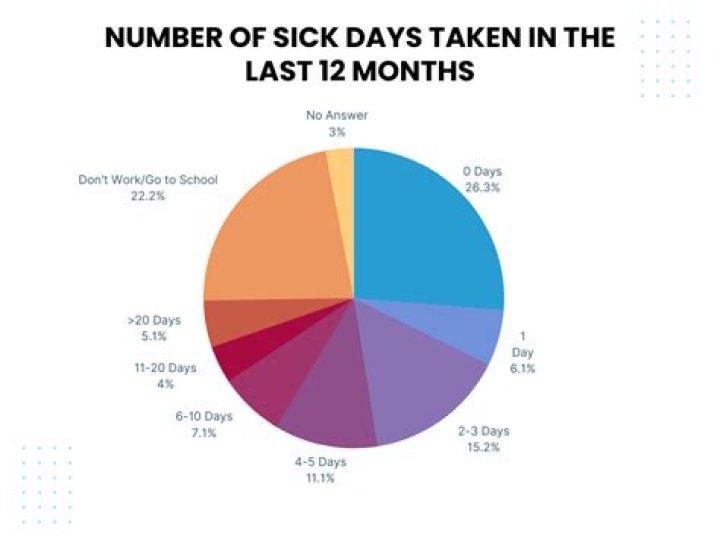
What is the difference between electrons in an orbit and in an orbital in the atomic model?
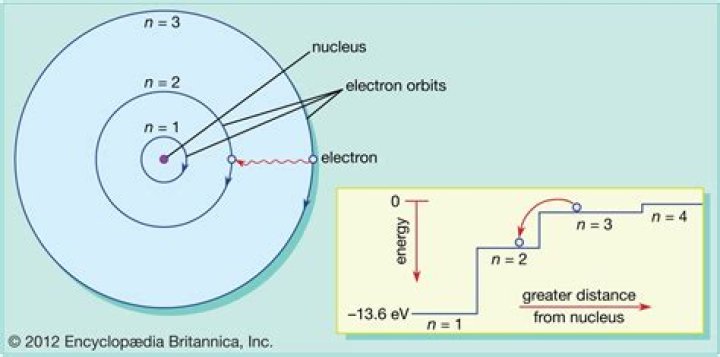
Atomic orbital is the wavefunction of the electron at a certain state determined by the quantum numbers. Electron orbit does not exist in Quantum Mechanics.
What is difference between orbital and subshell?
A subshell is composed of orbitals. The first shell consists of only one subshell that is ‘s’ and the second subshell consist of two subshells that is ‘s’ and ‘p’ and so on. Each subshell contains one or more orbitals. s contain one orbital, p contains 3 orbitals, d contains 5 orbitals and f contain 7 orbitals.
What is meant by orbit in chemistry?
An orbit in chemistry is the fixed path on which electron moves or revolves around the atom’s nucleus. Furthermore, an orbit is a simple planar representation of a particular electron.
What is the difference between its different orbits and how is the electron able to move from one to another?
Each orbital is at a different distances from the nucleus. Electrons in each orbital contain a set quantity of energy. As long as an electron remains in the same orbital, the energy content of that electron remains constant. Electrons can move between orbits by releasing or absorbing energy.
What is difference between Shell and orbit?
The main difference between shell subshell and orbital is that shells are composed of electrons that share the same principal quantum number and subshells are composed of electrons that share the same angular momentum quantum number whereas orbitals are composed of electrons that are in the same energy level but have …
What is difference between orbit and orbital in chemistry?
An orbit is a fixed path on which electrons revolve around the nucleus. An orbital is the probable area of finding the maximum density of electrons in an atom. An orbital is a three dimensional representation. An orbit is non-directional in nature which means the shape of an atom cannot be described by an orbit.
What is difference between shell and orbit?
How do electrons move between orbitals?
Electrons in each orbital contain a set quantity of energy. As long as an electron remains in the same orbital, the energy content of that electron remains constant. Electrons can move between orbits by releasing or absorbing energy.
How many orbitals are in a shell?
The number of orbitals in a shell is the square of the principal quantum number: 12 = 1, 22 = 4, 32 = 9. There is one orbital in an s subshell (l = 0), three orbitals in a p subshell (l = 1), and five orbitals in a d subshell (l = 2). The number of orbitals in a subshell is therefore 2(l) + 1.
What is the difference between orbitals and orbitals?
The major difference between orbit and orbitals is that the former is a definite path of electrons revolutions, whereas the latter represents the uncertain area with a high probability of finding an electron. An orbit in chemistry is a definite path whose shape is regular, on which revolution of electrons takes place.
What do you mean by Orbit?
A path on which electrons revolve around the nucleus of an atom is called Orbit. The three-dimensional space in which the chances of finding an electron is quite high is known as Orbital. It represents the exact position of an electron inside of an atom.
How many electrons can be in an orbit?
In an orbit number of electrons can be 2n2 where ‘n’ is number of orbit or principle quantum number. In one orbital maximum two electrons can be filled. Orbits are non-directional in nature. Orbitals are directional in nature except s-orbitals which are spherical in shape.
Why are orbitals non-directional in nature?
Orbits are non-directional in nature. Orbitals are directional in nature except s-orbitals which are spherical in shape. Orbits represent that position and momentum of an electron can be measured simultaneously with certainty. Which is against Heinsberg’s principle.