What is the coordination number of HCP?
12
Coordination number – the number of nearest neighbor atoms or ions surrounding an atom or ion. For FCC and HCP systems, the coordination number is 12.
What is the Z for HCP?
The value of Z is 6 because 6 is the coordination number of HCP…
How do you find the coordination number of a crystal structure?
It’s more difficult to determine chemical bonding in solid-state crystals, so the coordination number in crystals is found by counting the number of neighboring atoms. Most commonly, the coordination number looks at an atom in the interior of a lattice, with neighbors extending in all directions.
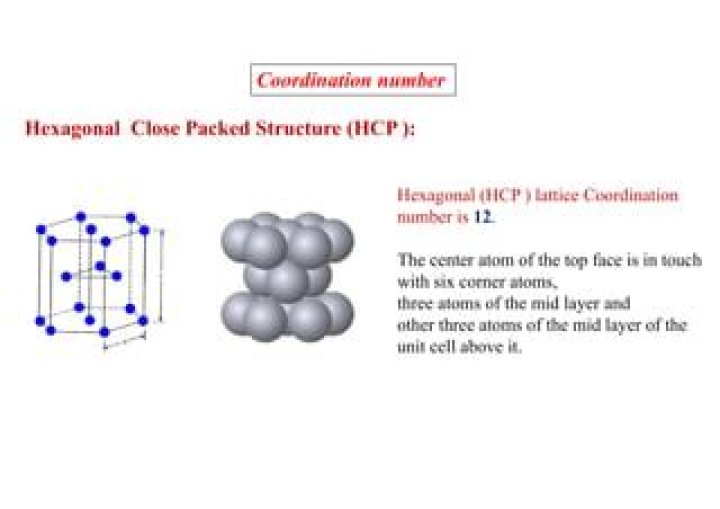
How do you find the coordination number of hexagonal close packing?
That can be simplified by saying that any given atom in hexagonal close packing is attached to 4 + 4 + 4 = 12 atoms at any point in time. hence by definition, the coordination number of hcp is 12.
How many atoms are there in hexagonal unit cell?
12 atoms
In HCP, there are 6 corner atoms in the top layer and 6 corner atoms in the bottom layer, so 12 atoms in the unit cell.
Which of the following has hexagonal crystal structure?
The metals that are a part of HCP crystal are Zinc, Magnesium, and Cadmium. While metals that are a part of CCP are, Copper, Silver, and Gold, which are coinage metals. Hence, magnesium, Mg is the metal that has HCP structure, so option B is correct.
What is the coordination number of hcp in three dimensions?
The coordination number of spheres in hcp lattice in three dimension is 6.
What is the coordination number of each atom in I hcp and II CCP?
Both in ccp and hcp structures, the coordination number is 12.
What is the coordination number in hexagonal close packed structure in two dimensions?
Each sphere is in contact with six of its neighbours and the two-dimensional coordination number is 6. The centres of these six spheres are at the corners of a regular hexagon hence this packing is called two-dimensional hexagonal close packing.
What is CN in hexagonal crystal structure?
Hexagonal Close-Packed Coordination Number. Coordination Number (CN) is the number of nearest neighbors that each atom has. As a close-packed structure, the HCP crystal has the number of nearest-neighbors (NN): 12. Each of these NNs contributes a bond, giving the crystal structure very high stability.
What is the coordination number of the hexagonal closest packed?
1 The hexagonal closest packed (hcp) has a coordination number of 12 and contains 6 atoms per unit cell. 2 The face-centered cubic (fcc) has a coordination number of 12 and contains 4 atoms per unit cell. 3 The body-centered cubic (bcc) has a coordination number of 8 and contains 2 atoms per unit cell.
What are the properties of a hexagonal closed packed crystal?
Here is a summary chart of all HCP crystal properties: Crystal Structure Hexagonal Close-Packed Atomic Packing Factor (APF) 74% Coordination Number 12 Number of Atoms per Unit Cell 6 Number of Octahedral Interstitial Sites 6
What is the coordination number of the crystal structure?
The Coordination number of the crystal structure is also called a ligancy number or ligancy. Co-ordination numbers of basic types of crystal structures are given below: Coordination Number of Simple Cubic Crystal Structure is 6 Coordination Number of Body Centered Crystal Structure (BCC) is 8

