What is normality Mohr salt?
The quantity of Mohr’s salt required for the 250ml of the solution having a normality of 0.05N can be calculated as follows. The molar mass of mohr’s salt = 392 g/mol. Strength = Normality x Equivalent weight. = (1/20) x 392 = 19.6 g/L. For preparing 250ml of N/20 Mohr’s salt solution, Mohr salt required.
What is Mohr salt experiment?
Mohr’s salt is prepared by adding a little amount of sulfuric acid in water and then adding an equimolar mixture of hydrated ferrous sulfate and ammonium sulfate in it. Now this resulting solution mixture is subjected to crystallization. After this we get light green coloured small crystals of Mohr’s salt.
How do you find the normality of Mohr salt?
The molar mass and equivalent mass of Mohr’s salt is same, 392g/mol. Oxidation of Mohr’s salt involves removal of one electron. So, concentration of Mohr’s salt in molarity and normality are the same.
Is Mohr salt secondary standard solution?
Oxalic acid, Mohr’s salt, potassium dichromate are some examples of primary standards.
What is formula of Mohr salt?
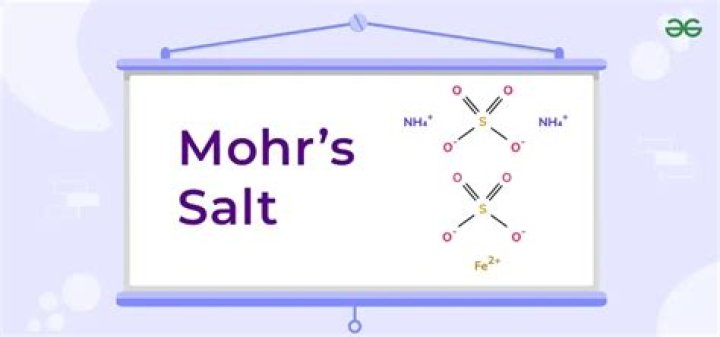
(NH₄)₂Fe(SO₄)₂·6H₂O
Ammonium iron(II) sulfate/Formula
What is the molarity of Mohr salt?
APPARATUS AND CHEMICALS REQUIRED– Mohr’s salt, weighing bottle, weight box, volumetric flask, funnel, distilled water, chemical balance, dilute H2SO4, beakers, conical flask, funnel, burette, pipette, clamp stand, tile, KMnO4 solution. Mohr’s salt having the formula FeSO4. (NH4)2SO4. 6H2O has molar mass 392gmol-1.
What is the molarity of Mohr’s salt?
What is the use of Mohr salt?
Mohr’s salt is used in analytical chemistry as the preferred source of ferrous ions due to the solid’s extended shelf life and resistance to oxidation. Sulfuric acid is often added to solutions using ferrous ammonium sulfate in order to reduce the oxidation of the ferric iron.
What is Mohr salt called?
Ammonium iron(II) sulfate
Ammonium iron(II) sulfate, or Mohr’s salt, is the inorganic compound with the formula (NH4)2Fe(SO4)2(H2O)6. Containing two different cations, Fe2+ and NH4+, it is classified as a double salt of ferrous sulfate and ammonium sulfate.
What type of salt is Mohr’s salt?
Ammonium iron(II) sulfate, or Mohr’s salt, is the inorganic compound with the formula (NH4)2Fe(SO4)2(H2O)6. Containing two different cations, Fe2+ and NH4+, it is classified as a double salt of ferrous sulfate and ammonium sulfate.
Why Mohr’s salt is called Mohr’s salt?
It can be noted that Mohr’s salt is named after the German scientist Karl Friedrich Mohr. When dissolved in water, Mohr’s salt (like most other salts of ferrous sulfate) dissolves to yield an aquo complex with the chemical formula [Fe(H2O)6]2+.
What type of salt is Mohr salt?
What is Mohr’s salt?
Mohr’s salt is a light green colour crystalline inorganic compound. It is a double salt of FeSO4 .7H2O and (NH4)2SO4 which belongs to a group of double sulfate named Schonites or tutton’s salts, { formula: M2M’ (SO4)2.6H2O where M is univalent and M’ is divalent ions. The chemical Mohr’s salt formula of the double salt is FeSO4.
What is the formula for ammonium iron sulfate (Mohr’s salt)?
Ammonium iron(II) sulfate, or Mohr’s salt, is the inorganic compound with the formula (NH 4) 2Fe(SO 4) 2·6H 2O.
What is the source of Fe2+ in Mohr salt?
In the field of analytical chemistry, Mohr’s salt is one of the most preferred compounds that acts as a source of Fe 2+ ions (or ferrous ions). The advantage of using this solid as a source of ferrous ions is that it has a relatively long shelf life and is also resistant to oxidation due to atmospheric exposure.
What is the normality of Mohr’s salt and KMnO4 solution?
N 1, N 2 are normality of Mohr’s salt and KMnO 4 solution respectively. V 1, V 2 are volume of Mohr’s salt and KMnO 4 respectively. The strength of and molarity of given KMnO 4 solution is found out as 2/y x 31.6 g/l and N/5 moles/liter, respectively.