What is Electrovalent bond with example?
An electrovalent bond is formed when a metal atom transfers one or more electrons to a non-metal atom. Some other examples are: MgCl2, CaCl2, MgO, Na2S, CaH2, AlF3, NaH, KH, K2O, KI, RbCl, NaBr, CaH2 etc.
What is an Electrovalent bond Class 10?
Electrovalent bonds are produced when electrons are transferred from atoms of one element to atoms of another element, producing positive and negative ions. The bond which is formed by the transfer of electrons between the atoms is called electrovalent bond or ionic bond.
What is Electrovalent bond explain with any two examples?
A chemical bond between two ions with opposite charges, characteristic of salts. Also called electrovalent bond . In sodium chloride, a sodium atom (Na) donates an electron to a chlorine atom (Cl), forming an ionic bond. Examples-MgCl2, CaCl2, MgO, Na2S, CaH2, AlF3, NaH, KH, K2O, KI, RbCl, NaBr, CaH2 etc.
What is the difference between Electrovalent bond and ionic bond?
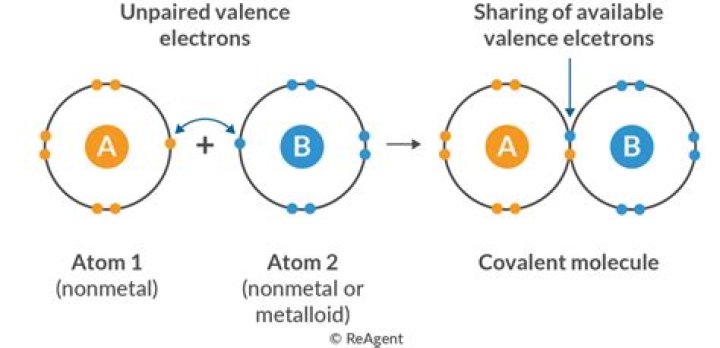
Electrovalent bond and covalent bond are two types of chemical bonds that are found between atoms of a molecule or a compound. Electrovalent bond is also called an ionic bond. It is an electrostatic attraction between two atoms. A covalent bond is a result of sharing electrons between two atoms.
What is another name for Electrovalent bond?
In this page you can discover 2 synonyms, antonyms, idiomatic expressions, and related words for electrovalent bond, like: ionic-bond and electrostatic bond.
Why is NaCl an Electrovalent bond?
Thus, one sodium atom gives away its one valence electron to the one chlorine atom and in this way, both the atoms complete their octet by attaining eight electrons in their outermost shells. Since NaCl compounds are also formed by the transfer of one electron thus, NaCl is an electrovalent compound.
Which is strongest bond in chemistry?
covalent bond
In chemistry, covalent bond is the strongest bond. In such bonding, each of two atoms shares electrons that binds them together. For example, water molecules are bonded together where both hydrogen atoms and oxygen atoms share electrons to form a covalent bond.
Is NaCl an Electrovalent bond?
Since NaCl compounds are also formed by the transfer of one electron thus, NaCl is an electrovalent compound. Hence, NaCl is an electrovalent compound.
How is Electrovalent bond formed?
Ionic bond, also called electrovalent bond, type of linkage formed from the electrostatic attraction between oppositely charged ions in a chemical compound. Such a bond forms when the valence (outermost) electrons of one atom are transferred permanently to another atom.
Which hydrogen bond is strongest?
As fluorine has small size and high electronegativity, it has high tendency to attract partial positive charge accumulated on H-atom. So, fluorine forms strongest H-bond.
What is the strongest bond in the universe?
The strongest bond in the universe!
| Property (CCSD/aug-cc-pVTZ) | N≡N | (H-N≡N-H)2+ |
|---|---|---|
| NN stretch, cm-1 | 2418.8 | 2226.3/3024.0 2688.4a/2567.7b |
| ELF NN basin integration | 3.57 | 4.59 |
| QTAIM ρ(r)/∇2ρ(r) | 0.714/-3.38 | 0.700/-2.96 |
| aValue for hydrogen mass of 10,000 bValue for hydrogen mass of 0.001. |
What’s the difference between Electrovalent and covalent bond?
1 ) Electrovalent compounds are formed by complete rtransfer of electrons while covalent compounds are formed by sharing of electrons between 2 atoms. 2) Electrovalent compounds are more soluble in polar solvents like water while covalent compounds are more soluble in non polar solvents like methane.
Which covalent bond is the strongest?
sigma bond
A sigma bond is the strongest type of covalent bond, in which the atomic orbitals directly overlap between the nuclei of two atoms. Sigma bonds can occur between any kind of atomic orbitals; the only requirement is that the atomic orbital overlap happens directly between the nuclei of atoms.
What is the weakest bond?
Explanation: A Chemical bond is technically a bond between two atoms that results in the formation of a molecule , unit formula or polyatomic ion. The weakest of the intramolecular bonds or chemical bonds is the ionic bond. The ionic bond is generally the weakest of the true chemical bonds that bind atoms to atoms.
What is the weakest hydrogen bond?
The weakest hydrogen bonds considered in the literature are about 0.5 kcal/mol. Most of hydrogen-bonded complexes of interest form the group of moderate hydrogen bonds. Water dimer or hydrogen fluoride dimer are typical examples for this group.
What is Electrovalent bond and covalent bond?
Electrovalent bond is a chemical bond between two atoms due to a transfer of electron(s) from one atom to the other. Covalent bond is a type of chemical bond which occurs due to sharing of electron pairs between atoms.
It has seven electrons in its outermost shell or chlorine has 7 valence electrons. Since NaCl compounds are also formed by the transfer of one electron thus, NaCl is an electrovalent compound. Hence, NaCl is an electrovalent compound.
Which one is the strongest bond?
In chemistry, covalent bond is the strongest bond. In such bonding, each of two atoms shares electrons that binds them together.
What is the definition of an electrovalent bond?
electrovalent bond. n. (Chemistry) a type of chemical bond in which one atom loses an electron to form a positive ion and the other atom gains the electron to form a negative ion.
What are the properties of an electrovalent compound?
Characteristics of Electrovalent Compounds. Crystal Structure: In the solid state of electrovalent compounds, anions and cations are arranged in a regular manner. This is a crystal in which anions are surrounded by a definite number of cations and vice-versa.
What kind of bond is formed when valence electron is transferred?
The bond formed by this kind of combination is an ionic bond or electrovalent bond. An Ionic bond is the bond formed by the complete transfer of valence electron so as to attain stability. This type of bonding leads to the formation of two oppositely charged ions.
How is the electrovalent bond between sodium and chlorine formed?
For example, the bond between the sodium and chlorine atoms in sodium chloride (NaCl) is formed by the transfer of an electron from sodium to chlorine, creating Na + and Cl – ions. The electrostatic attraction between these ions provides the bonding in NaCl. Pick a style below, and copy the text for your bibliography. ” electrovalent bond .

