What is complex compound in chemistry?
-A complex compound can be described as a class of substances with a chemical structure where the central atom is a metal and it is surrounded by non-metal atoms or a group of atoms (or ligands). – Complex compound is also called a coordinate compound as it contains the coordinate bonds in its structure.
What are the examples of complexes?
The Naming of Complexes
| Examples in Which the Complex Is Cation | |
|---|---|
| [Co(NH3)6]Cl3 | hexaaminecobalt(III) chloride |
| [Pt(NH3)2Cl4] | diaminetetrachloroplatinum(IV) |
| [Ni(H2NCH2CH2NH2)2Cl2] | dichlorobis(ethylenediamine)nickel(II) |
| Examples in Which the Complex Is an Anion |
What is complex chemistry example?
Another Example of a Complex An example is the tetrachlorocobaltate complex anion, whose formula is [CoCl4]2- (see image). The cobalt is in oxidation state 2, binding with four chloride ions. Each one of the chloride ions brings one negative charge to the complex, so the total charge on the complex is -2.
What is the Colour of complex compound?
The energy corresponding to the wavelength will impart colour to that compound. And if the electron does not make transition , there will be no colour i.e. the substance will be colourless. The colour of the compound is due to the d−d transition of the electrons. Hence, the correct option is (C).
What is a complex ionic compound?
A complex ion has a metal ion at its center with a number of other molecules or ions surrounding it. These can be considered to be attached to the central ion by coordinate (dative covalent) bonds (although in some cases, the bonding is actually more complicated than that.)
What is meant by complex compound give two examples?
Examples of complex compound include potassium ferrocyanide K4[Fe(CN)6] and potassium ferricyanide K3[Fe(CN)6]. Other examples include pentaamine chloro cobalt(III) chloride [Co(NH)5Cl]Cl2 and dichlorobis (ethylenediammine) platinum(IV) nitrate [Pt(en)2Cl2](NO3)2.
How do you name a complex compound?
When naming a complex ion, the ligands are named before the metal ion. Write the names of the ligands in the following order: neutral, negative, positive. If there are multiple ligands of the same charge type, they are named in alphabetical order. (Numerical prefixes do not affect the order.)
How complex compounds are formed?
A complex ion is a species formed between a central metal ion and one or more surrounding ligands, molecules or ions that contain at least one lone pair of electrons. Small, highly charged metal ions have the greatest tendency to act as Lewis acids and form complex ions.
Which is the example of complex compound?
That is, it contains at least two independent clauses (like a compound sentence) and at least one dependent clause (like a complex sentence). For example: Erin loves her brother, and he loves her too because she pays his bills. The dog ran off when I chased him, but I didn’t care.
How complex compound is formed?
How do complexes appear Colourful?
When ligands attach to a transition metal to form a coordination complex, electrons in the d orbital split into high energy and low energy orbitals. As certain wavelengths are absorbed in this process, subtractive color mixing occurs and the coordination complex solution becomes colored.
What is ligand class 12?
Ligands. The atoms or groups which are attached directly to central atoms are called ligands. Ligands are Lewis bases which donates electron pair and forms coordinate bonds with the metal atom. For example: H2O, CO, NO2‒, etc. A ligand may be neutral, positively or negatively charged.
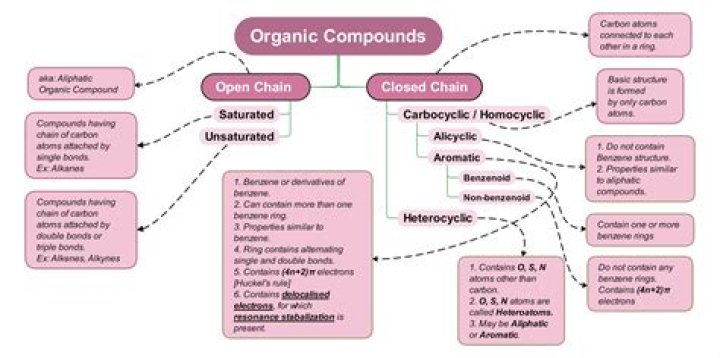
What are the most common organic compounds?
Acetone. A rather potent chemical,acetone is found in common products such as nail polish remover,furniture polish and wallpaper.
What are the rules for naming organic compounds?
The steps for naming an organic compound are: Identification of the parent hydrocarbon chain. Identification of the parent functional group, if any, with the highest order of precedence. Identification of the side-chains.
What is the name of the compound CH3?
Butyraldehyde, also known as butanal, is an organic compound with the formula CH3(CH2)2CHO. This compound is the aldehyde derivative of butane. It is a colourless flammable liquid with an acrid smell.