What are the 4 types of thermodynamic process explain each?
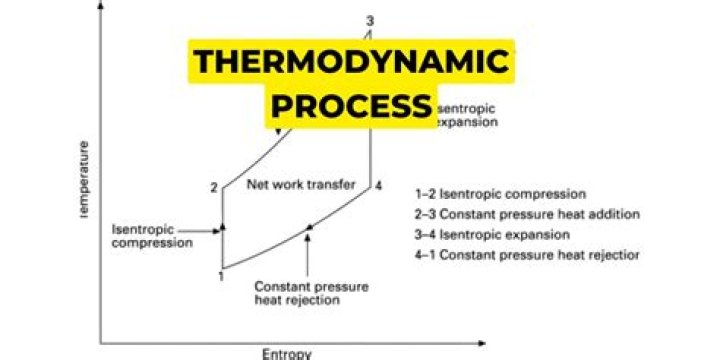
The four types of thermodynamic process are isobaric, isochoric, isothermal and adiabatic.
What are the different types of thermodynamic process?
There are several types of thermodynamic processes, including (a) isothermal, where the system’s temperature is constant; (b) adiabatic, where no heat is exchanged by the system; (c) isobaric, where the system’s pressure is constant; and (d) isochoric, where the system’s volume is constant.
What are thermodynamic processes explain?
Thermodynamic Processes A thermodynamic process is defined as a change from one equilibrium macrostate to another macrostate. The initial and final states are the defining elements of the process. In this process energy may be transferred form or into the system and also work can be done by or on the system.
What is isotropic in thermodynamics?
Isotropic systems are uniform regardless of direction. Radiation may be isotropic when the the observed intensity is the same in all directions from the source. A fluid may be called isotropic if the relationships between stress and rate of strain is the same in all directions.
What is thermodynamic process Class 12?
A process by which two or more state variables of a system can be changed is called a thermodynamic process.
How many processes are there in thermodynamics?
Classical thermodynamics considers three main kinds of thermodynamic process: (1) changes in a system, (2) cycles in a system, and (3) flow processes. (1) A change in a system is defined by a passage from an initial to a final state of thermodynamic equilibrium.
Why do we read thermodynamics?
Thermodynamics is a very important branch of both physics and chemistry. It deals with the study of energy, the conversion of energy between different forms and the ability of energy to do work.
What is isotherm Class 11?
An isothermal process is one in which the temperature of a system stays constant. That is, ΔT=0 An isotherm is a type of curve. An isotherm is a line drawn on a map or chart that connects points of equal temperature. That is, the temperature values are the same at any point along an isotherm.
What is isothermal process in thermodynamics class 11?
Isothermal Process: An isothermal process may be defined as a thermodynamic process in which the temperature remains constant. All the processes which are taking place inside a refrigerator are isothermal processes as there is no change in its internal temperature.
What is the meaning of isotropic process?
In thermodynamics, an isentropic process is an idealized thermodynamic process that is both adiabatic and reversible. It means a process in which the entropy of the system remains unchanged; as mentioned, this could occur if the process is both adiabatic and reversible.
What is difference between homogeneous and isotropic material?
Difference Between Homogeneous and Isotropic Definition. Homogeneous: Homogeneous refers to the uniformity of the structure of matter. Structure. Homogeneous: The structure of homogeneous material is uniform. Direction. Homogeneous: The properties of homogeneous matter does not depend on the direction. Examples. Conclusion.
Is a reversible process isentropic?
1 Answer 1. Taken from wikipedia: In thermodynamics , an isentropic process is an idealized thermodynamic process that is adiabatic and in which the work transfers of the system are frictionless; there is no transfer of matter and the process is reversible. An isentropic process is, by definition, adiabatic and reversible.
Is an isentropic process adiabatic?
In thermodynamics , an isentropic process is an idealized thermodynamic process that is both adiabatic and reversible. The work transfers of the system are frictionless, and there is no transfer of heat or matter.
What does isentropic flow mean?
Isentropic flow. In fluid dynamics, an isentropic flow is a fluid flow that is both adiabatic and reversible. That is, no heat is added to the flow, and no energy transformations occur due to friction or dissipative effects.