Is myosin powered by ATP hydrolysis?
The “power stroke” mechanism for myosin movement along actin filaments: The direction in which the actin filament will be moved is dictated by the structural orientation of myosin in relation to the filament. A complete round of ATP hydrolysis produces a single ‘step’ or movement of myosin along the actin filament.
Does actin produce myosin and ATP?
Actin filaments, usually in association with myosin, are responsible for many types of cell movements. Myosin is the prototype of a molecular motor—a protein that converts chemical energy in the form of ATP to mechanical energy, thus generating force and movement.
Do proteins hydrolyze ATP?
Biomolecular motor proteins like myosin generate mechanical force from the chemical energy of ATP. Like gas engines, they have different parts (protein domains) that run through a well-defined cycle of motions, consuming one ATP per cycle. Because ATP is very stable, motor proteins catalyze its breakdown (hydrolysis).
What are the 3 roles of ATP in muscle contraction?
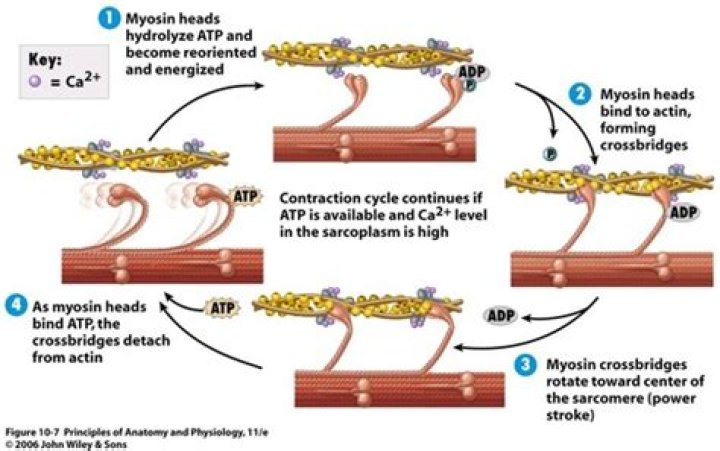
It also reminds us that ATP is needed by the muscle cell for the power stroke of the myosin cross bridge, for disconnecting the cross bridge from the binding site on actin, and for transporting calcium ions back into the SR.
Why does myosin bind to actin?
Myosin has another binding site for ATP at which enzymatic activity hydrolyzes ATP to ADP, releasing an inorganic phosphate molecule and energy. ATP binding causes myosin to release actin, allowing actin and myosin to detach from each other.
How do myosin and actin muscles respond to ATP?
When calcium ions are added, they combine with troponin, inhibition is released, actin and myosin interact, and ATP is broken down. In smooth muscles, Ca2+ activates an enzyme (kinase) that catalyzes the transfer of phosphate from ATP to myosin, and the phosphorylated form is then activated by actin.
How does ATP affect the relationship between actin and myosin?
Myosin binds to actin at a binding site on the globular actin protein. Myosin has another binding site for ATP at which enzymatic activity hydrolyzes ATP to ADP, releasing an inorganic phosphate molecule and energy. ATP binding causes myosin to release actin, allowing actin and myosin to detach from each other.
What happens in ATP hydrolysis?
ATP hydrolysis is the catabolic reaction process by which chemical energy that has been stored in the high-energy phosphoanhydride bonds in adenosine triphosphate (ATP) is released after splitting these bonds, for example in muscles, by producing work in the form of mechanical energy.
What is the delta G for ATP hydrolysis?
-57 kJ/mol
ATP is hydrolyzed to ADP in the reaction ATP+H2O→ADP+Pi+ free energy; the calculated ∆G for the hydrolysis of 1 mole of ATP is -57 kJ/mol.
What happens to myosin when ATP is hydrolyzed?
The ATP is hydrolyzed into ADP and inorganic phosphate (P i) by the enzyme ATPase. The energy released during ATP hydrolysis changes the angle of the myosin head into a “cocked” position, ready to bind to actin if the sites are available. ADP and Pi remain attached; myosin is in its high energy configuration.
How does ataTP prepare myosin for binding with actin?
ATP prepares myosin for binding with actin by moving it to a higher- energy state and a “cocked” position. Once the myosin forms a cross-bridge with actin, the Pi disassociates and the myosin undergoes the power stroke, reaching a lower energy state when the sarcomere shortens.
How does ATP hydrolysis move the actin filament?
T The direction in which the actin filament will be moved is dictated by the structural orientation of myosin in relation to the filament. A complete round of ATP hydrolysis produces a single ‘step’ or movement of myosin along the actin filament.
How do myosin motor proteins work?
Each myosin motor protein possesses ATPase activity and functions in a cyclical manner that couples ATP binding and hydrolysis to a conformational change in the protein. This process is known as the ‘powerstroke cycle’ (reviewed in [1][2][3]) and is outlined in the steps below using myosin II as an example.

