Is freezing an exothermic or endothermic reaction?
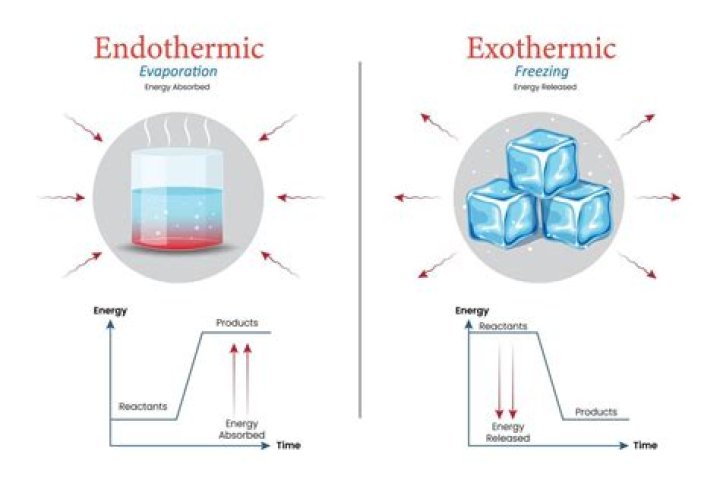
When water becomes a solid, it releases heat, warming up its surroundings. This makes freezing an exothermic reaction. One common endothermic reaction is ice melting.
Are acid reactions exothermic or endothermic?
Acid base neutralization involves the formation of a salt and water. Such a process is inevitably exothermic.
Why is freezing an endothermic reaction?
As aresult, the frezing of water is an exothermic process because heat is being removed from the system. Melting ice is an endothermic process because you need to provide heat in order to allow the molecules to overpower the hydrogen bonds and start moving about again.
Are base endothermic or exothermic?
In fact strong acid-base reactions are typically exothermic, meaning that the new bond formed between the proton (H+) and the base is stronger than the bond that was broken to release the H+. The released energy raises the temperature of the surroundings.
Can freezing be endothermic?
Fusion, vaporization, and sublimation are endothermic processes, whereas freezing, condensation, and deposition are exothermic processes.
Is freezing point exothermic?
In this process of freezing, water loses heat to the surroundings, so it is an exothermic process.
Is acid base reaction neutralization?
A neutralization reaction is when an acid and a base react to form water and a salt and involves the combination of H+ ions and OH- ions to generate water. The neutralization of a strong acid and strong base has a pH equal to 7.
Why is acid base neutralization reactions exothermic?
SInce strong acids and strong bases are completely dissociated in solution, no formal bonds are being broken. The formation of two very strong covalent bonds between hydrogen and the hydroxide ion is responsible for the neutralization reaction’s exothermic character.
Why is acid base Neutralisation exothermic?
Why is freezing an exothermic reaction?
When the water is placed in a freezer, the water slowly loses heat to the surrounding cold air. The water molecules on losing energy begin to move slowly, come closer and pack close enough to change to ice. In this process, the water releases heat to the surroundings, so it is an exothermic process.
Is the melting of ice exothermic or endothermic?
Endothermic Reactions The input of heat energy, from the surroundings, triggers the breakdown of chemical and physical bonds. One of the most common endothermic reactions is the melting of ice.
Does freezing absorb or release heat?
Note that melting and vaporization are endothermic processes in that they absorb or require energy, while freezing and condensation are exothermic process as they release energy.
Are acid base reactions endothermic or exothermic?
Acid-base reactions are exothermic. However, part of the difference between a strong acid and a weak acid is the amount of heat they release when reacting with a strong base (like KOH) and a weak base (like NH4OH).
Is bond-breaking endothermic or exothermic?
Explanation: Bond-breaking is endothermic, whereas bond formation is exothermic. Whether a given reaction is exothermic or endothermic depends on the balance between bond-breaking and bond-making. In the generalized acid base reaction, as shown below, we form 2 substances, salt and water, whose formation should be intrinsically exothermic:
Is adding baking soda to vinegar endothermic or exothermic?
Neutralization reactions (acid + base = salt + water) are usually exothermic but when you add baking soda to vinegar, or HCl it is slightly endothermic. The neutralization reaction actually does release heat: Energy absorbed by products leaving +HC2H3O2 + NaHCO3 CO2 + NaC2H3O2 (aq) + H2O + Energy released due to bond formation
What is an example of an exothermic reaction?
In the generalized acid base reaction, as shown below, we form 2 substances, salt and water, whose formation should be intrinsically exothermic: And for an obvious example… H Cl(aq) + N aOH (aq) acid + base → N aCl(aq) + H 2O(l) salt + water

