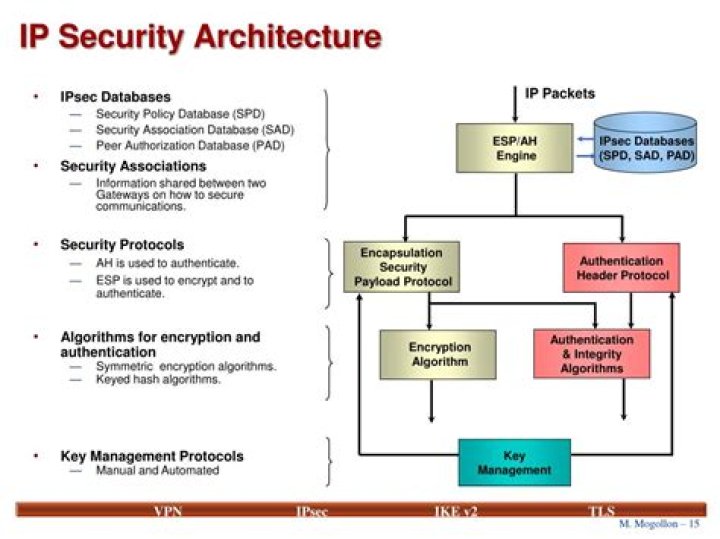
How is Charles law used in experiments?
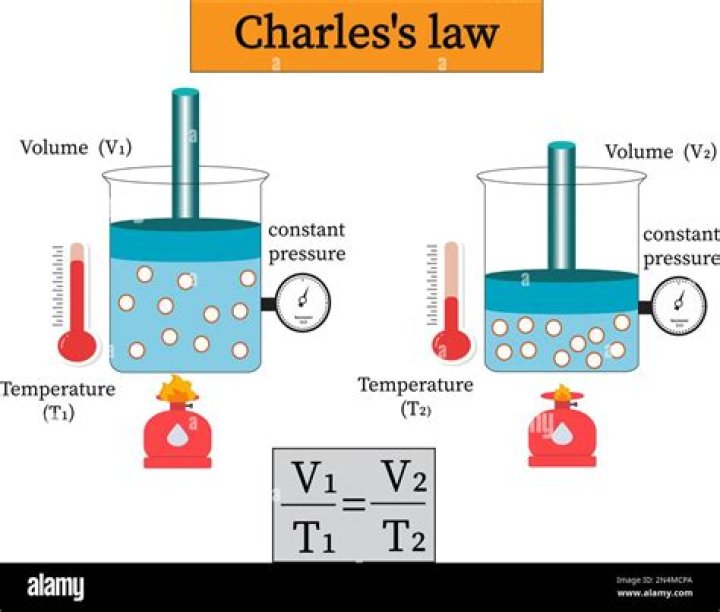
The equation for Charles’s law can be expressed as V1/T1=V2/T2. In other words, if a balloon is filled with air, it will shrink if cooled and expand if heated. This happens because the air inside the balloon, which is a gas, takes up a smaller volume when it is cool, and takes up a larger volume when it is heated.
How do you record the volume of an air in an Erlenmeyer flask?
Double-click on the Erlenmeyer flask to access the options to open or close the flask and label your flask with a new name. Choose the option to close the flask and click Done. An airtight stopper is now placed into the mouth of the flask, sealing the air inside. Record the volume of air in the flask.
Why was the flask held in the boiling water for 5 minutes?
Keep the flask immersed in the water for at least 5 min. This will allow the gas on the inside of the flask to attain the temperature of the surrounding water. While the gas cools, it will decrease in volume, sucking water into the flask.
What experiment did Charles do?
Charles’s law Around 1787 Charles did an experiment where he filled 5 balloons to the same volume with different gases. He then raised the temperature of the balloons to 80 °C (not at constant temperature) and noticed that they all increased in volume by the same amount.
How do you investigate Charles Law?
Charles’s Law
- Heating a container filled with a mass of gas.
- To investigate the relationship between volume and temperature, at constant pressure, an experiment can be carried out where a fixed mass of gas is in a container, which is free to expand, without any gas escaping.
How do you find Charles Law?
Based on the definition of Charles’ law, we can write the Charles’ law equation in the following way: V₁ / T₁ = V₂ / T₂ , where V₁ and T₁ are initial volume and temperature, respectively. Similarly, V₂ and T₂ are the final values of these gas parameters.
How do you explain Charles Law?
Charles’s law, a statement that the volume occupied by a fixed amount of gas is directly proportional to its absolute temperature, if the pressure remains constant. It is a special case of the general gas law and can be derived from the kinetic theory of gases under the assumption of a perfect (ideal) gas.
What is the purpose of filling the flask with water up to its brim?
It is reached by diluting a known quantity of the investigated solution in a conical flask, adding distilled water to the mark to it. This allows you to determine the exact concentration of the test solution and determine the accuracy of the study by conducting a series of similar experiments.
What is experimental verification of Charles Law?
Experimental Verification of Charles’ Law: The given mass of a gas is enclosed in the cylinder. Temperature of the gas is increased by heating which causes to increase the volume. By changing the temperature at constant pressure, different volumes are obtained. V/T is always found constant at the same pressure.
How do you use an Erlenmeyer flask?
Put about 200 mL of water into a second 250 mL Erlenmeyer flask (Flask no. 2) and place the end of the rubber tubing into the water. Make sure that the end of the rubber tubing reaches to the bottom of the flask and stays submerged at all times.
What is V1 and V2 in a conical flask?
V1 is the volume of the air in the flask at the boiling point of the water bath. t2 is the temperature of the air when the flask is submerged in the water bath. Vw is the volume of the water moved in the flask. V2 is the volume of the air at temperature t2. Thoroughly clean the conical flask with a paper towel.
How do you measure the temperature of water in a flask?
Place the thermometer in the beaker to measure the temperature of the water. Once the temperature crosses 95 °C, the water is about to start boiling. Let the heating continue for 6-7 min more. We want the air inside the flask to be at the same temperature as of the boiling water.
How does Charles’s law relate volume to temperature?
Charles’s law relates volume to temperature at a constant pressure. Thus, in the experiment below, we will be studying volume versus temperature relationship under a constant pressure. The experiment is performed at a constant atmospheric pressure. The experiment proceeds by placing an empty flask in a boiling water bath.