How does temperature affect Bohr effect?
The CO, Bohr factor is increased at high temperatures; this is primarily due to increased carbamino formation with rising temperature, especially at lower oxygen saturation. The effect of DPG on oxygen affinity is reduced at a high temperature and elevated at low temperature.
What is the Bohr shift effect?
The Bohr effect is the shift to the right of the oxygen equilibrium curve of both adult and fetal blood in response to an increase in PCO2 or a decrease in pH, or both.
How is the Bohr shift a result of a change in the environment?
That is, the Bohr effect refers to the shift in the oxygen dissociation curve caused by changes in the concentration of carbon dioxide or the pH of the environment. Conversely, a decrease in carbon dioxide provokes an increase in pH, which results in hemoglobin picking up more oxygen.
How does temperature impact oxygen-hemoglobin dissociation?
A higher temperature is correlated to the cells working harder and therefore means they need a higher supply of oxygen to keep them going. Therefore, as temperature increases, this shifts the entire oxygen-hemoglobin dissociation curve to the right.
How does temperature affect oxygen transport?
Increased temperatures of blood result in a reduced affinity of hemoglobin for oxygen and thus a rightward shift of the Oxygen-Hemoglobin Dissociation Curve described in Oxygen Transport. Consequently, higher temperatures result in enhanced unloading of oxygen by hemoglobin.
How does temperature affect oxygen concentration?
Air and Water Temperature Increases Lower levels of dissolved oxygen due to the inverse relationship that exists between dissolved oxygen and temperature. As the temperature of the water increases, dissolved oxygen levels decrease.
What is the Bohr shift a level PE?
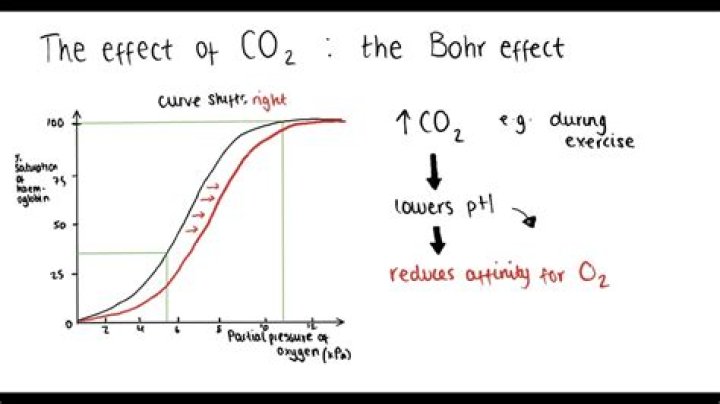
The Bohr Shift describes the movement of the oxygen dissociation curve to the right of normal. This occurs due to increased levels of carbon dioxide, such as when a person increases their exercise level, which causes an increased concentration of carbonic acid to be formed.
What is Bohr and Haldane effect?
“The Bohr-Haldane effect is a physicochemical phenomenon which describes the changes in affinity for nonoxygen ligand binding by haemoglobin which result from the conformal changes induced in the haemoglobin tetramer by the binding of oxygen to haem” However, that does not exactly roll off the tongue.
Why is deoxygenated hemoglobin Favoured by low pH?
The formation of this bond stabilises the deoxygenated T-state; on the other hand oxyhaemoglobin cannot form this sort of bond and is unaffected by pH. Thus, as soon as oxyhaemoglobin releases its O2 cargo, the presence of a low pH locks it in an unreceptive state, preventing it from binding oxygen molecules.
How does pH affect oxygen affinity?
Oxygen Transport As blood nears the lungs, the carbon dioxide concentration decreases, causing an increase in pH. This increase in pH increases hemoglobin’s affinity for oxygen through the Bohr effect, causing hemoglobin to pick up oxygen entering your blood from your lungs so it can transport it to your tissues.
How does temperature affect oxygen loading and unloading?
How do pH and temperature affect the oxygen Haemoglobin dissociation curve?
With increased carbon dioxide excretion, increased hydrogen ion (proton, H+) concentration (fall in pH) and increased partial temperature, the oxygen dissociation curve is shifted to the right, promoting oxygen dissociation. At this time, the affinity of hemoglobin for oxygen (P50) becomes large.
What is Bohr shift or Bohr effect?
Changes in the oxygen dissociation curve as a result of carbon dioxide levels are known as the Bohr shift or Bohr effect When the partial pressure of carbon dioxide is high, in respiring tissues for example, haemoglobin’s affinity for oxygen is reduced
What is the Bohr effect in hypoxia?
Bohr Effect. Chronic hypoxia increases the blood s concentration of 2,3-DPG which also shifts the curve to the right. The presence of HbF and carbon monoxide (CO) shift the curve to the left, increasing the oxygen affinity of hemoglobin.
What is the Bohr coefficient of oxygen delivery?
Lapennas determined that the optimal Bohr coefficient for O 2 delivery under steady-state conditions (and with many assumptions) is 0.5 × RQ. This represents a compromise between pH sensitivity of the Hb and the resulting pH change that occurs during capillary blood transit.
How do you find the Bohr effect strength formula?
The formula is obtained by plotting the logarithm of this measurement on a graph at various pH levels, yielding a line with slope equal to . Bohr effect strength exhibits an inverse relationship with the size of an organism: the magnitude increases as size and weight decreases.

