How do you write a clinical trial protocol?
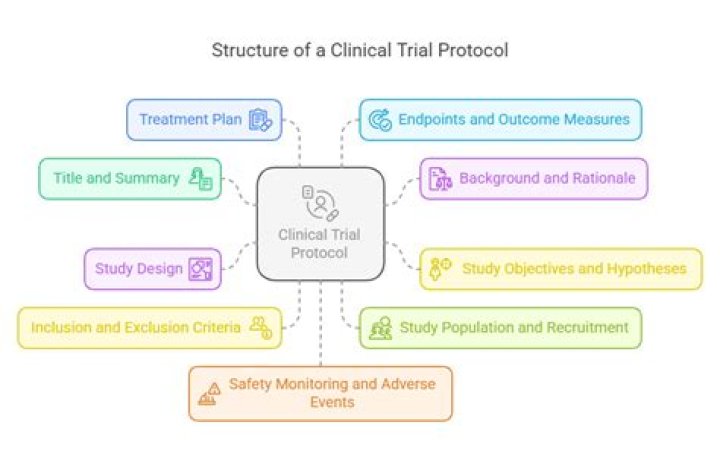
According to the ICH Good Clinical Practice guidelines, a protocol should include the following topics:
- Title Page (General Information)
- Background Information.
- Objectives/Purpose.
- Study Design.
- Selection and Exclusion of Subjects.
- Treatment of Subjects.
- Assessment of Efficacy.
- Assessment of Safety.
What are the key elements of a clinical study protocol?
we can read, includes: General information, background information, objectives / purpose, trial design , selections and withdrawal of subjects, treatment subjects, assessment of safety and efficacy.
How do you write a good study protocol?
The protocol should outline the rationale for the study, its objective, the methodology used and how the data will be managed and analysed. It should highlight how ethical issues have been considered, and, where appropriate, how gender issues are being addressed.
Who designs clinical trial protocol?
The actual writing of a protocol is a team effort with contributions from a medical expert, a statistician, a pharmacokinetics expert, the clinical research coordinator, and the project manager, who all provide input to the medical writer to produce the final document.
What is the most appropriate content in a clinical trial protocol?
Including name and description of the investigational product(s); a summary of findings from non-clinical and other clinical studies; known and potential risks and benefits; description of route of administration and dosage; statement ensuring compliance with protocol, GCP and regulatory requirements; description of …
Who writes a clinical trial protocol?
How do I create a medical protocol?
- Developing a Protocol.
- GENERAL PROTOCOL CHECKLIST.
- Title. Protocol summary.
- Literature review/current state of knowledge about project topic. Justification for study.
- DESIGN. How study design or surveillance system addresses hypotheses and.
- VARIABLES/INTERVENTIONS. Variables.
- DATA HANDLING AND. ANALYSIS.
- HANDLING OF UNEXPECTED.
Who prepares and approves the study protocol?
45 CFR 46 requires that a committee, at the NIH and elsewhere called an Institutional Review Board, or IRB, prospectively and regularly review and approve human-subjects research activities. The primary mandate of the IRB is to protect and safeguard the rights and welfare of human subjects.
What is a protocol template?
The Protocol Template for Behavioral and Social Sciences Research is a resource for communicating the science, methods, and operations of a clinical trial. This template is a suggested format for clinical trials that are testing a behavioral or social intervention or experimental manipulation.
How to be part of a clinical trial?
Ask your doctor for a referral. Getting a referral from your primary healthcare provider to participate in the clinical trial may be the quickest and easiest way for you
How to find a clinical trial?
Gather Details about Your Cancer. If you decide to look for a clinical trial,you will need to know certain details about your cancer diagnosis and compare these
What must a clinical trial involve?
Clinical trials aim to find out whether a medical strategy, treatment, or device is safe and effective for humans to use or consume. Trials consist of four phases, and they can focus on: treatment, prevention, diagnostic, screening, supportive care, health services research, and basic science.

