How do you draw an atom of an element?
Starts here5:12How to Draw an Atom! – YouTubeYouTubeStart of suggested clipEnd of suggested clip51 second suggested clipThis is called the nucleus. And it’s where most of the atoms mass is located the electrons goMoreThis is called the nucleus. And it’s where most of the atoms mass is located the electrons go outside the nucleus. But.
What is an example of an atom of an element?
Hydrogen (H2), oxygen (O2), and chlorine (Cl2) molecules, for example, each contains two atoms. Another form of oxygen, ozone (O3), has three atoms, and sulfur (S8) has eight atoms. All elemental molecules are made of atoms of a single element. Fig.
What are 10 examples of atoms?
Here are some examples of atoms:
- Neon (Ne)
- Hydrogen (H)
- Argon (Ar)
- Iron (Fe)
- Calcium (Ca)
- Deuterium, an isotope of hydrogen that has one proton and one neutron.
- Plutonium (Pu)
- F-, a fluorine anion.
How do you draw elements?
Starts here10:21How to draw an atom – YouTubeYouTube
How do you draw and label atoms?
Starts here5:17Chemistry: Draw and Label an Atom – YouTubeYouTube
What are 5 elements examples?
Common examples of elements are iron, copper, silver, gold, hydrogen, carbon, nitrogen, and oxygen.
What is atom and example of atom?
Many atoms consist of a positively charged nucleus consisting of protons and neutrons surrounded by a cloud of electrons charged negatively. An atom is any particle of matter at its most basic level which contains at least one proton. Here are some examples of the atoms: hydrogen (H) neon (Ne).
What is an atom diagram?
Atomic diagrams were developed to explain the interaction of the elements of the Earth and space long before atoms could be observed. Nowadays, scientists can see particles that are smaller than an atom. These sub-atomic particles are the basis of particle physics.
What is an example of element?
An element would have the same number of protons in their atomic nuclei. For example, carbon is an element comprised of atoms having the same number of protons, i.e. 6. Common examples of elements are iron, copper, silver, gold, hydrogen, carbon, nitrogen, and oxygen.
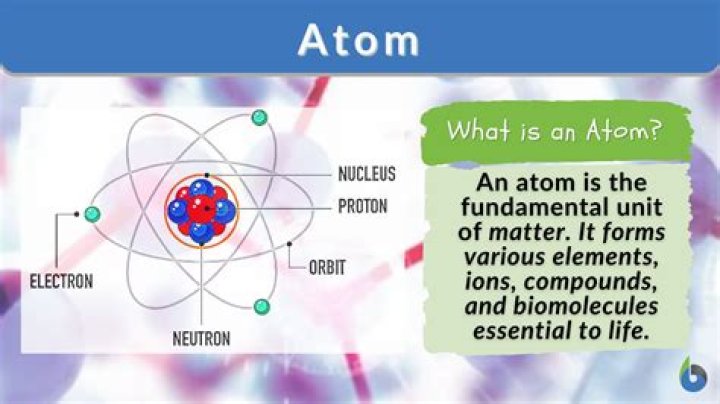
What is the structure of an atom?
Picture given below shows, structure of atom and locations of proton neutron and electron in atom. Center of the atom, including protons and neutrons, is called also nucleus and circles around nucleus are called orbits.
What are some examples of elements and their symbols?
20 Examples of Elements and Their Symbols. Chemical elements are known by their names and their symbols. Examples include hydrogen, helium, carbon, and nitrogen. Chemical elements are known by their names and their symbols. Examples include hydrogen, helium, carbon, and nitrogen. Menu.
Can you work out the link between an atom’s electronic structure and period?
Look at the atoms drawn below and see if you can work out the link between an atom’s electronic structure, its group and its period: The number of electron shells an element possess is the same as its period. Have a look at the diagram above – hydrogen and helium are in period 1 and they have one electron shell.
How do you read the periodic table of an atom?
Atoms and the Periodic Table. The atomic number indicates how many protons there are in the nucleus of an atom. The atomic mass tells us how many protons and neutrons are in the nucleus. The number of electrons are the same as the number of protons in a neutral atom. The vertical columns are known as periodic table groups.

