Does protein maintain acid-base balance?
Protein is also essential in maintaining proper pH balance (the measure of how acidic or basic a substance is) in the blood.
How does protein affect acid-base balance?
Further analyses of the interrelation between diet and acid-base status revealed that increasing protein intake (despite its potential to increase NAE) also significantly improves the capacity for renal net acid excretion by stimulating urinary ammonium excretion.
How do proteins help maintain fluid and acid-base balance?
Fluid balance – Proteins regulate body processes to maintain fluid balance. Proteins in the blood are called albumin and globulin, and they help maintain the body’s fluid balance by keeping water in the blood. Blood proteins have the ability to attract and keep fluid in the bloodstream.
Why does protein cause acidosis?
It is well known that a high-protein diet induces metabolic acidosis due to acidic residues of proteins. Metabolic acidosis induced by high dietary protein increases urinary acid excretion and also increases urinary calcium and phosphate levels, which may negatively influence bone and muscle protein metabolism.
Which two body systems contribute to acid-base balance?
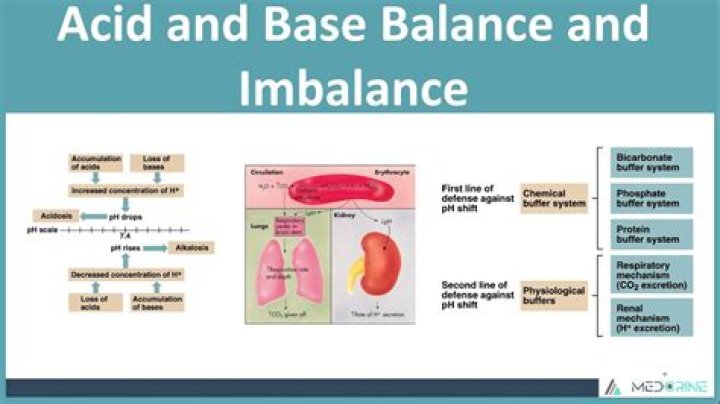
Your kidneys and lungs work to maintain the acid-base balance. Even slight variations from the normal range can have significant effects on your vital organs. Acid and alkaline levels are measured on a pH scale.
How does protein maintain pH?
One way your body regulates pH is with proteins. An example is hemoglobin, a protein that makes up red blood cells. Hemoglobin binds small amounts of acid, helping to maintain the normal pH value of your blood. The other buffer systems in your body include phosphate and bicarbonate ( 16 ).
How do proteins contribute to acid-base balance of the blood quizlet?
how do proteins contribute to acid-base balance of the blood? pump ions into and out of cells and serve as buffers that resists changes in pH.
Why is acid-base balance important in food?
The acid-base balance is essential for the smooth operation of the body’s metabolic functions. We often hear about the ideal pH value (7.36 to 7.44), which is especially important in the blood. The blood pH value is thus in the slightly basic range.
What is protein balance and why is it important?
Every cell in the human body contains protein. The basic structure of protein is a chain of amino acids. You need protein in your diet to help your body repair cells and make new ones. Protein is also important for growth and development in children, teens, and pregnant women.
How does protein maintain pH balance?
Does protein catabolism cause metabolic acidosis?
Metabolic acidosis increases protein degradation resulting in muscle wasting and a negative nitrogen balance. The branched-chain amino acids serve as useful markers of these changes and their catabolism is increased in acidosis, particularly for the spontaneous acidosis associated with renal failure.
Does high protein cause acidosis?
Background: High protein diet has been known to cause metabolic acidosis, which is manifested by increased urinary excretion of nitrogen and calcium. Bodybuilders habitually consumed excessive dietary protein over the amounts recommended for them to promote muscle mass accretion.
What is the clinical significance of acid-base balance?
Acid-base balance in the human body is one of the most paramount physiological processes. The clinical significance of acid-base balance is one which is hard to deny. Some of the most common admissions to hospitals are due to diseases that can dangerously affect the acid-base balance.
How does the renal system regulate the body’s acid-base balance?
The renal regulation of the body’s acid-base balance addresses the metabolic component of the buffering system. Whereas the respiratory system (together with breathing centers in the brain) controls the blood levels of carbonic acid by controlling the exhalation of CO 2, the renal system controls the blood levels of bicarbonate.
Do low-carbohydrate high-protein diets affect acid-base balance and bone metabolism?
Background: Low-carbohydrate high-protein (LCHP) diets are used commonly for weight reduction. This study explores the relationship between such diets and acid-base balance, kidney-stone risk, and calcium and bone metabolism. Methods: Ten healthy subjects participated in a metabolic study.
How is carbon dioxide instrumental to acid-base balance in the body?
This carbon dioxide is instrumental to acid-base balance in the body which is demonstrated with the following reaction: CO2 + H20 <-> H2C03 <-> HCO3- + H+ The carbon dioxide formed during cellular respiration combines with water to create carbonic acid. Carbonic acid then dissociates into bicarbonate and a hydrogen ion.